Research reveals how irisin—a hormone produced during endurance exercise—works in multiple ways to protect brain health and prevent Alzheimer’s and dementia in mammalian models https://t.co/WExA9ZUVZN via @HarvardMagazine pic.twitter.com/WnLw3iWDPe
— Harvard Medical School (@harvardmed) December 22, 2021
Tag Archives: Exercise Studies
Exercise: The Evolution Of Human Physical Activity
Human physical activities differ significantly from other species. How, when and why did these capabilities evolve? What adaptations underlie them? And how did the evolution of human physical activity affect other key human characteristics that have advanced our species?
Herman Pontzer explores the evolution of human metabolism and its role in our evolution and health. From an evolutionary perspective, life is a game of turning energy into offspring. The strategies that species use to acquire energy, in the form of food, and allocate energy to the essential tasks of growth, maintenance, movement, and reproduction, are incredibly diverse and reflect the ecological pressures and opportunities encountered. There is a deep evolutionary history of the human metabolic strategy and our divergence from other apes.
Timeline: 00:00 – Start 01:38 – The Evolution of Human Metabolism
More from: CARTA: The Evolution of Human Physical Activity (https://www.uctv.tv/carta-physical-ac…)
Infographic: Exercise For ‘Claudication’ (BMJ Study)
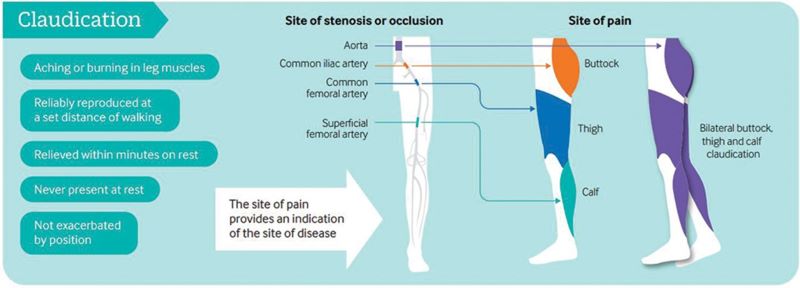
Exercise training is a safe, effective and low-cost intervention for improving walking ability in patients with IC. Additional benefits may include improvements in QoL, muscle strength and cardiorespiratory fitness. Clinical guidelines advocate supervised exercise training as a primary therapy for IC, with walking as the primary modality.
However, evidence is emerging for the role of various other modes of exercise including cycling and progressive resistance training to supplement walking training. In addition, there is emerging evidence for home-based exercise programmes. Revascularisation or drug treatment options should only be considered in patients if exercise training provides insufficient symptomatic relief.
Abstract
Peripheral artery disease (PAD) is caused by atherosclerotic narrowing of the arteries supplying the lower limbs often resulting in intermittent claudication, evident as pain or cramping while walking. Supervised exercise training elicits clinically meaningful benefits in walking ability and quality of life. Walking is the modality of exercise with the strongest evidence and is recommended in several national and international guidelines. Alternate forms of exercise such as upper- or lower-body cycling may be used, if required by certain patients, although there is less evidence for these types of programmes. The evidence for progressive resistance training is growing and patients can also engage in strength-based training alongside a walking programme. For those unable to attend a supervised class (strongest evidence), home-based or ‘self-facilitated’ exercise programmes are known to improve walking distance when compared to simple advice. All exercise programmes, independent of the mode of delivery, should be progressive and individually prescribed where possible, considering disease severity, comorbidities and initial exercise capacity. All patients should aim to accumulate at least 30 min of aerobic activity, at least three times a week, for at least 3 months, ideally in the form of walking exercise to near-maximal claudication pain.
Covid-19 Infographic: ‘Exercise Is Medicine’ (BMJ)


There are over 35,000,000 reported cases of COVID-19 disease and 1 000 000 deaths across more than 200 countries worldwide.1 With cases continuing to rise and a robust vaccine not yet available for safe and widespread delivery, lifestyle adaptations will be needed for the foreseeable future. As we try to contain the spread of the virus, adults are spending more time at home. Recent evidence2 suggests that physical activity levels have decreased by ~30% and sitting time has increased by ~30%. This is a major concern as physical inactivity and sedentary behaviour are risk factors3 for cardiovascular disease, obesity, cancer, diabetes, hypertension, bone and joint disease, depression and premature death.
To date, more than 130 authors from across the world have provided COVID-19-related commentary on these concerns. Many experts4 have emphasised the importance of increasing healthy living behaviours and others5 have indicated that we are now simultaneously fighting not one but two pandemics (ie, COVID-19, physical inactivity). Physical inactivity alone results in over 3 million deaths per year5 and a global burden of US$50 billion.6 Immediate action is required to facilitate physical activity during the COVID-19 pandemic because it is an effective form of medicine3 to promote good health, prevent disease and bolster immune function. Accordingly, widespread messaging to keep adults physically active is of paramount importance.
Several organisations including the WHO, American Heart Association and American College of Sports Medicine have offered initial suggestions and resources for engaging in physical activity during the COVID-19 pandemic. Expanding on these resources, our infographic aims to present a comprehensive illustration for promoting daily physical activity to the lay audience during the COVID-19 pandemic (figure 1). As illustrated, adults are spending more time at home, moving less and sitting more. Physical activity provides numerous health benefits, some of which may even help directly combat the effects of COVID-19. For substantial health benefits, adults should engage in 150–300 min of moderate-to-vigorous intensity physical activity each week and limit the time spent sitting. The recommended levels of physical activity are safely attainable even at home. Using a combination of both formal and informal activities, 150 min can be reached during the week with frequent sessions of physical activity spread throughout the day. Sedentary behaviour can be further reduced by breaking up prolonged sitting with short active breaks. In summary, this infographic offers as an evidence-based tool for public health officials, clinicians, educators and policymakers to communicate the importance of engaging in physical activity during the COVID-19 pandemic.
Studies: Osteoarthritis Patients Using Exercise Therapy Have Less Pain, Cut Opioid & Analgesic Use


Conclusion Among patients with knee or hip OA using analgesics, more than half either discontinued analgesic use or shifted to lower risk analgesics following an 8-week structured exercise therapy and patient education programme (GLA:D). These data encourage randomised controlled trial evaluation of whether supervised exercise therapy, combined with patient education, can reduce analgesic use, including opioids, among patients with knee and hip OA pain.
Study: “Intensive Diet And Exercise” Reverses Type 2 Diabetes In 61% Of Patients
From The Lancet Diabetes & Endocrinology (June 2020):
Our findings show that the intensive lifestyle intervention led to significant weight loss at 12 months, and was associated with diabetes remission in over 60% of participants and normoglycaemia in over 30% of participants. The provision of this lifestyle intervention could allow a large proportion of young individuals with early diabetes to achieve improvements in key cardiometabolic outcomes, with potential long-term benefits for health and wellbeing.

Type 2 diabetes is affecting people at an increasingly younger age, particularly in the Middle East and in north Africa. We aimed to assess whether an intensive lifestyle intervention would lead to significant weight loss and improved glycaemia in young individuals with early diabetes.
.
Between July 16, 2017, and Sept 30, 2018, we enrolled and randomly assigned 158 participants (n=79 in each group) to the study. 147 participants (70 in the intervention group and 77 in the control group) were included in the final intention-to-treat analysis population. Between baseline and 12 months, the mean bodyweight of participants in the intervention group reduced by 11·98 kg (95% CI 9·72 to 14·23) compared with 3·98 kg (2·78 to 5·18) in the control group (adjusted mean difference −6·08 kg [95% CI −8·37 to −3·79], p<0·0001). In the intervention group, 21% of participants achieved more than 15% weight loss between baseline and 12 months compared with 1% of participants in the control group (p<0·0001). Diabetes remission occurred in 61% of participants in the intervention group compared with 12% of those in the control group (odds ratio [OR] 12·03 [95% CI 5·17 to 28·03], p<0·0001). 33% of participants in the intervention group had normoglycaemia compared with 4% of participants in the control group (OR 12·07 [3·43 to 42·45], p<0·0001).
.
Health: Parkinson’s Disease And The “Overall Benefits Of Exercise”
Exercise: Older Adults With Higher Muscle Mass Reduce Cardiovascular Disease By Over 80% (Study)
From a Journal of Epidemiology & Community Health online release:
 The 10 year CVD (cardiovascular disease) incidence increased significantly across the baseline SMI (skeletal muscle mass index) tertiles (p<0.001). Baseline SMM (Skeletal muscle mass) showed a significant inverse association with the 10 year CVD incidence (HR 0.06, 95% CI 0.005 to 0.78), even after adjusting for various confounders. Additionally, participants in the highest SMM tertile had 81% (95% CI 0.04 to 0.85) lower risk for a CVD event as compared with those in the lowest SMM tertile.
The 10 year CVD (cardiovascular disease) incidence increased significantly across the baseline SMI (skeletal muscle mass index) tertiles (p<0.001). Baseline SMM (Skeletal muscle mass) showed a significant inverse association with the 10 year CVD incidence (HR 0.06, 95% CI 0.005 to 0.78), even after adjusting for various confounders. Additionally, participants in the highest SMM tertile had 81% (95% CI 0.04 to 0.85) lower risk for a CVD event as compared with those in the lowest SMM tertile.
Background Skeletal muscle mass (SMM) is inversely associated with cardiometabolic health and the ageing process. The aim of the present work was to evaluate the relation between SMM and 10 year cardiovascular disease (CVD) incidence, among CVD-free adults 45+ years old.

Methods ATTICA is a prospective, population-based study that recruited 3042 adults without pre-existing CVD from the Greek general population (Caucasians; age ≥18 years; 1514 men). The 10 year study follow-up (2011–2012) captured the fatal/non-fatal CVD incidence in 2020 participants (50% men). The working sample consisted of 1019 participants, 45+ years old (men: n=534; women: n=485). A skeletal muscle mass index (SMI) was created to reflect SMM, using appendicular skeletal muscle mass (ASM) standardised by body mass index (BMI). ASM and SMI were calculated with specific indirect population formulas.
The global population is ageing at an unprecedented speed, especially in Europe. As a concept, ageing is considered a continuous process starting from birth and is accompanied by various physiological changes and a number of comorbidities1 2 that affect health and quality of life.3 4 Skeletal muscle mass (SMM)4 alterations are among these physiological changes.
SMM tissue decline, as a part of these physiological changes, starts in middle age (or even earlier, in the 30s) and progresses in more advanced age.5 It has been shown that SMM declines with a rate of more than 3% per decade starting from the age of 30+.6 Half of the human body’s mass is actually SMM and it has an active role in numerous metabolic pathways.5 7 SMM decline is related, among others, to various disability patterns, poor mental health and increased mortality.5 7 8 In addition, well documented studies have shown that SMM alterations are related to cardiovascular health,9 10 even different muscle morphology.11 Recently, Srikanthan et al 12 reported the importance of muscle tissue in relation to cardiovascular disease (CVD) and total mortality in stable CVD patients.
Infographic: Exercise For Intermittent Leg Pain & Walking (Claudication)

Health Studies: Vigorous Daily Exercise Increases Neurotropins, Boosting Cognitive Function
From the Journal of Sport and Health Science:
Therefore, promotion of adequate volumes and intensities of physical exercise (i.e., approximately 3 months of moderate-intensity aerobic exercise, with 2–3 sessions/week lasting not less than 30 min) represents an inexpensive and safe strategy for boosting BDNF (brain-derived neurotrophic factor) release that may preserve or restore cognitive function.

Taken together, the currently available data seemingly confirm the existence of a positive relationship between physical exercise and circulating BDNF levels, both in the short and long term, and also support the beneficial impact of training programs for amplifying the acute BDNF response.