How a Texas robot named Apollo became a meditation on dignity, dependence, and the future of care.

This essay is inspired by an episode of the WSJ Bold Names podcast (September 26, 2025), in which Christopher Mims and Tim Higgins speak with Jeff Cardenas, CEO of Apptronik. While the podcast traces Apollo’s business and technical promise, this meditation follows the deeper question at the heart of humanoid robotics: what does it mean to delegate dignity itself?
By Michael Cummins, Editor, September 26, 2025
The robot stands motionless in a bright Austin lab, catching the fluorescence the way bone catches light in an X-ray—white, clinical, unblinking. Human-height, five foot eight, a little more than a hundred and fifty pounds, all clean lines and exposed joints. What matters is not the size. What matters is the task.
An engineer wheels over a geriatric training mannequin—slack limbs, paper skin, the posture of someone who has spent too many days watching the ceiling. With a gesture the engineer has practiced until it feels like superstition, he cues the robot forward.
Apollo bends.
The motors don’t roar; they murmur, like a refrigerator. A camera blinks; a wrist pivots. Aluminum fingers spread, hesitate, then—lightly, so lightly—close around the mannequin’s forearm. The lift is almost slow enough to be reverent. Apollo steadies the spine, tips the chin, makes a shelf of its palm for the tremor the mannequin doesn’t have but real people do. This is not warehouse choreography—no pallets, no conveyor belts. This is rehearsal for something harder: the geometry of tenderness.
If the mannequin stays upright, the room exhales. If Apollo’s grasp has that elusive quality—control without clench—there’s a hush you wouldn’t expect in a lab. The hush is not triumph. It is reckoning: the movement from factory floor to bedside, from productivity to intimacy, from the public square to the room where the curtains are drawn and a person is trying, stubbornly, not to be embarrassed.
Apptronik calls this horizon “assistive care.” The phrase is both clinical and audacious. It’s the third act in a rollout that starts in logistics, passes through healthcare, and ends—if it ever ends—at the bedroom door. You do not get to a sentence like that by accident. You get there because someone keeps repeating the same word until it stops sounding sentimental and starts sounding like strategy: dignity.
Jeff Cardenas is the one who says it most. He moves quickly when he talks, as if there are only so many breaths before the demo window closes, but the word slows him. Dignity. He says it with the persistence of an engineer and the stubbornness of a grandson. Both of his grandfathers were war heroes, the kind of men who could tie a rope with their eyes closed and a hand in a sling. For years they didn’t need anyone. Then, in their final seasons, they needed everyone. The bathroom became a negotiation. A shirt, an adversary. “To watch proud men forced into total dependency,” he says, “was to watch their dignity collapse.”
A robot, he thinks, can give some of that back. No sigh at 3 a.m. No opinion about the smell of a body that has been ill for too long. No making a nurse late for the next room. The machine has no ego. It does not collect small resentments. It will never tell a friend over coffee what it had to do for you. If dignity is partly autonomy, the argument goes, then autonomy might be partly engineered.
There is, of course, a domestic irony humming in the background. The week Cardenas was scheduled to sit for an interview about a future of household humanoids, a human arrived in his own household ahead of schedule: a baby girl. Two creations, two needs. One cries, one hums. One exhausts you into sleeplessness; the other promises to be tireless so you can rest. Perhaps that tension—between what we make and who we make—is the essay we keep writing in every age. It is, at minimum, the ethical prompt for the engineering to follow.
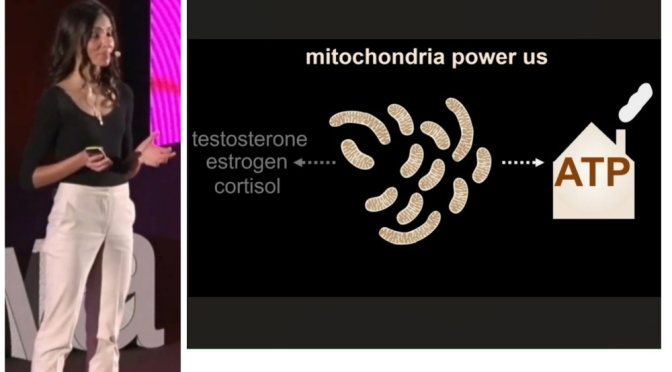
In the lab, empathy is equipment. Apollo’s body is a lattice of proprietary actuators—the muscles—and a tangle of sensors—the nerves. Cameras for eyes, force feedback in the hands, gyros whispering balance, accelerometers keeping score of every tilt. The old robots were position robots: go here, stop there, open, close, repeat until someone hit the red button. Apollo lives in a different grammar. It isn’t memorizing a path through space; it’s listening, constantly, to the body it carries and the moment it enters. It can’t afford to be brittle. Brittleness drops the cup. And the patient.
But muscle and nerve require a brain, and for that Apptronik has made a pragmatic peace with the present: Google DeepMind is the partner for the mind. A decade ago, “humanoid” was a dirty word in Mountain View—too soon, too much. Now the bet is that a robot shaped like us can learn from us, not only in principle but in practice. Generative AI, so adept at turning words into words and images into images, now tries to learn movement by watching. Show it a person steadying a frail arm. Show it again. Give it the perspective of a sensor array; let it taste gravity through a gyroscope. The hope is that the skill transfers. The hope is that the world’s largest training set—human life—can be translated into action without scripts.
This is where the prose threatens to float away on its own optimism, and where Apptronik pulls it back with a price. Less than a luxury car, they say. Under $50,000, once the supply chain exists. They like first principles—aluminum is cheap, and there are only a few hundred dollars of it in the frame. Batteries have ridden down the cost curve on the back of cars; motors rode it down on the back of drones. The math is meant to short-circuit disbelief: compassion at scale is not only possible; it may be affordable.
Not today. Today, Apollo earns its keep in the places compassion is an accounting line: warehouses and factories. The partners—GXO, Mercedes—sound like waypoints on the long gray bridge to the bedside. If the robot can move boxes without breaking a wrist, maybe it can later move a human without breaking trust. The lab keeps its metaphors comforting: a pianist running scales before attempting the nocturne. Still, the nocturne is the point.
What changes when the machine crosses a threshold and the space smells like hand soap and evening soup? Warehouse floors are taped and square; homes are not. Homes are improvisations of furniture and mood and politics. The job shifts from lifting to witnessing. A perfect employee becomes a perfect observer. Cameras are not “eyes” in a home; they are records. To invite a machine into a room is to invite a log of the room. The promise of dignity—the mercy of not asking another person to do what shames you—meets the chill of being watched perfectly.
“Trust is the long-term battle,” Cardenas says, not as a slogan but like someone naming the boss level in a game with only one life. Companies have slogans about privacy. People have rules: who gets a key, who knows where the blanket is. Does a robot get a key? Does it remember where you hide the letter from the old friend? The engineers will answer, rightly, that these are solvable problems—air-gapped systems, on-device processing, audit logs. The heart will answer, not wrongly, that solvable is not the same as solved.
Then there is the bigger shadow. Cardenas calls humanoid robotics “the space race of our time,” and the analogy is less breathless than it sounds. Space wasn’t about stars; it was about order. The Moon was a stage for policy. In this script the rocket is a humanoid—replicable labor, general-purpose motion—and the nation that deploys a million of them first rewrites the math of productivity. China has poured capital into robotics; some of its companies share data and designs in a way U.S. rivals—each a separate species in a crowded ecosystem—do not. One country is trying to build a forest; the other, a bouquet. The metaphor is unfair and therefore, in the compressed logic of arguments, persuasive.
He reduces it to a line that is either obvious or terrifying. What is an economy? Productivity per person. Change the number of productive units and you change the economy. If a robot is, in practice, a unit, it will be counted. That doesn’t make it a citizen. It makes it a denominator. And once it’s in the denominator, it is in the policy.
This is the point where the skeptic clears his throat. We have heard this promise before—in the eighties, the nineties, the 2000s. We have seen Optimus and its cousins, and the men who owned them. We know the edited video, the cropped wire, the demo that never leaves the demo. We know how stubborn carpets can be and how doors, innocent as they seem, have a way of humiliating machines.
The lab knows this better than anyone. On the third lift of the morning, Apollo’s wrist overshoots with a faint metallic snap, the servo stuttering as it corrects. The mannequin’s elbow jerks, too quick, and an engineer’s breath catches in the silence. A tiny tweak. Again. “Yes,” someone says, almost to avoid saying “please.” Again.
What keeps the room honest is not the demo. It’s the memory you carry into it. Everyone has one: a grandmother who insisted she didn’t need help until she slid to the kitchen floor and refused to call it a fall; a father who couldn’t stand the indignity of a hand on his waistband; the friend who became a quiet inventory of what he could no longer do alone. The argument for a robot at the bedside lives in those rooms—in the hour when help is heavy and kindness is too human to be invisible.
But dignity is a duet word. It means independence. It also means being treated like a person. A perfect lift that leaves you feeling handled may be less dignified than an imperfect lift performed by a nurse who knows your dog’s name and laughs at your old jokes. Some people will choose privacy over presence every time. Others want the tremor in the human hand because it’s a sign that someone is afraid to hurt them. There is a universe of ethics in that tremor.
The money is not bashful about picking a side. Investors like markets that look like graphs and revolutions that can be amortized—unlike a nurse’s memory of the patient who loved a certain song, which lingers, resists, refuses to be tallied. If a robot can deliver the “last great service”—to borrow a phrase from a theologian who wasn’t thinking of robots—it will attract capital because the service can be repeated without running out of love, patience, or hours. The price point matters not only because it makes the machine seem plausible in a catalog but because it promises a shift in who gets help. A family that cannot afford round-the-clock care might afford a tireless assistant for the night shift. The machine will not call in sick. It will not gossip. It will not quit. It will, of course, fail, and those failures will be as intimate as its successes.
There are imaginable safeguards. A local brain that forgets what it doesn’t need to know. A green light you can see when the camera is on. Clear policies about where data goes and who can ask for it and how long it lives. An emergency override you can use without being a systems administrator at three in the morning. None of these will quiet the unease entirely. Unease is the tax we pay for bringing a new witness into the house.
And yet—watch closely—the room keeps coaching the robot toward a kind of grace. Engineers insist this isn’t poetry; it’s control theory. They talk about torque and closed loops and compliance control, about the way a hand can be strong by being soft. But if you mute the jargon, you hear something else: a search for a tempo that reads as care. The difference between a shove and a support is partly physics and partly music. A breath between actions signals attention. A tiny pause at the top of the lift says: I am with you. Apollo cannot mean that. But it can perform it. When it does, the engineers get quiet in the way people do in chapels and concert halls, the secular places where we admit that precision can pass for grace and that grace is, occasionally, a kind of precision.
There is an old superstition in technology: every new machine arrives with a mirror for the person who fears it most. The mirror in this lab shows two figures. In the first: a patient who would rather accept the cold touch of aluminum than the pity of a stranger. In the second: a nurse who knows that skill is not love but that love, in her line of work, often sounds like skill. The mirror does not choose. It simply refuses to lie.
The machine will steady a trembling arm, and we will learn a new word for the mix of gratitude and suspicion that touches the back of the neck when help arrives without a heartbeat. It is the geometry of tenderness, rendered in aluminum. A question with hands.
THIS ESSAY WAS WRITTEN AND EDITED UTILIZING AI