By Michael Cummins, Editor, August 6, 2025

The theatrical landscapes of England and France, while both flourishing in the early modern period, developed along distinct trajectories, reflecting their unique cultural, philosophical, and political climates. The English Renaissance stage, exemplified by the towering figures of Christopher Marlowe and William Shakespeare, embraced a sprawling, often chaotic, exploration of human experience, driven by individual ambition and psychological depth. In contrast, the French Neoclassical theatre, championed by masters like Molière and Jean Racine, championed order, reason, and a more focused examination of societal manners and tragic passions within a stricter dramatic framework.
This essay will compare and contrast these two powerful traditions by examining how Marlowe and Shakespeare’s expansive and character-driven dramas differ from Molière’s incisive social comedies and Racine’s intense psychological tragedies. Through this comparison, we can illuminate the divergent artistic philosophies and societal preoccupations that shaped the dramatic arts in these two influential European nations.
English Renaissance Drama: The Expansive Human Spirit and Societal Flux
The English Renaissance theatre was characterized by its boundless energy, its disregard for classical unities, and its profound interest in the multifaceted human psyche. Playwrights like Christopher Marlowe and William Shakespeare captured the era’s spirit of exploration and individualism, often placing ambitious, flawed, and deeply introspective characters at the heart of their narratives. These plays, performed in bustling public theaters, offered a mirror to an English society grappling with rapid change, shifting hierarchies, and the exhilarating—and terrifying—potential of the individual.
Christopher Marlowe (1564–1593), a contemporary and rival of Shakespeare, pioneered the use of blank verse and brought a new intensity to the English stage. His plays often feature protagonists driven by overwhelming, almost superhuman, desires—for power, knowledge, or wealth—who challenge societal and divine limits. In Tamburlaine the Great, the Scythian shepherd rises to conquer empires through sheer force of will, embodying a ruthless individualism that defied traditional hierarchies. Marlowe’s characters are often defined by their singular, often transgressive, ambition.
“I hold the Fates bound fast in iron chains, / And with my hand turn Fortune’s wheel about.” — Christopher Marlowe, Tamburlaine the Great
Similarly, Doctor Faustus explores the dangerous pursuit of forbidden knowledge, with its protagonist selling his soul for intellectual mastery and worldly pleasure. Marlowe’s drama is characterized by its grand scale, its focus on the exceptional individual, and its willingness to delve into morally ambiguous territory, reflecting a society grappling with new ideas about human potential and the limits of authority. His plays were often spectacles of ambition and downfall, designed to provoke and awe, suggesting an English fascination with the raw, unbridled power of the individual, even when it leads to destruction. They spoke to a society where social mobility, though limited, was a potent fantasy, and where traditional religious and political certainties were increasingly open to radical questioning.
William Shakespeare (1564–1616) built upon Marlowe’s innovations, expanding the scope of English drama to encompass an unparalleled range of human experience. While his historical plays and comedies are diverse, his tragedies, in particular, showcase a profound psychological realism. Characters like Hamlet, Othello, and King Lear are not merely driven by singular ambitions but are complex individuals wrestling with internal conflicts, moral dilemmas, and the unpredictable nature of fate. Shakespeare’s plays often embrace multiple plots, shifts in tone, and a blend of prose and verse, reflecting the messy, unconstrained reality of life.
“All the world’s a stage, / And all the men and women merely players; / They have their exits and their entrances; / And one man in his time plays many parts…” — William Shakespeare, As You Like It
Hamlet’s introspection and indecision, Lear’s descent into madness, and Othello’s tragic jealousy reveal a deep fascination with the inner workings of the human mind and the devastating consequences of human fallibility. Unlike the French emphasis on decorum, Shakespeare’s stage could accommodate violence, madness, and the full spectrum of human emotion, often without strict adherence to classical unities of time, place, or action. This freedom allowed for a rich, multifaceted exploration of the human condition, making his plays enduring studies of the soul. These plays vividly portray an English society grappling with the breakdown of traditional order, the anxieties of political succession, and the moral ambiguities of power. They suggest a national character more comfortable with contradiction and chaos, finding truth in the raw, unfiltered experience of human suffering and triumph rather than in neat, rational resolutions.
French Neoclassical Drama: Order, Reason, and Social Control
The French Neoclassical theatre, emerging in the 17th century, was a reaction against the perceived excesses of earlier drama, favoring instead a strict adherence to classical rules derived from Aristotle and Horace. Emphasizing reason, decorum, and moral instruction, playwrights like Molière and Jean Racine crafted works that were elegant, concentrated, and deeply analytical of human behavior within a structured society. These plays offered a reflection of French society under the centralized power of the monarchy, particularly the court of Louis XIV, where order, hierarchy, and the maintenance of social appearances were paramount.
Molière (Jean-Baptiste Poquelin, 1622–1673), the master of French comedy, used wit and satire to expose the follies, hypocrisies, and social pretensions of his contemporary Parisian society. His plays, such as Tartuffe, The Misanthrope, and The Miser, feature characters consumed by a single dominant passion or vice (e.g., religious hypocrisy, misanthropy, avarice). Molière’s genius lay in his ability to create universal types, using laughter to critique societal norms and encourage moral rectitude. His comedies often end with the restoration of social order and the triumph of common sense over absurdity.
“To live without loving is not really to live.” — Molière, The Misanthrope
Unlike the English focus on individual transformation, Molière’s characters often remain stubbornly fixed in their vices, serving as satirical mirrors for the audience. The plots are tightly constructed, adhering to the classical unities, and the language is precise, elegant, and witty, reflecting the French emphasis on clarity and rational thought. His plays were designed not just to entertain, but to instruct and reform, making them crucial vehicles for social commentary. Molière’s comedies reveal a French society deeply concerned with social decorum, the perils of pretense, and the importance of maintaining a rational, harmonious social fabric. They highlight the anxieties of social climbing and the rigid expectations placed upon individuals within a highly stratified and centralized court culture.
Jean Racine (1639–1699), the preeminent tragedian of the French Neoclassical period, explored the destructive power of human passions within a highly constrained and formal dramatic structure. His tragedies, including Phèdre, Andromaque, and Britannicus, focus intensely on a single, overwhelming emotion—often forbidden love, jealousy, or ambition—that inexorably leads to the protagonist’s downfall. Racine’s plays are characterized by their psychological intensity, their elegant and precise Alexandrine verse, and their strict adherence to the three unities (time, place, and action).
“There is no greater torment than to be consumed by a secret.” — Jean Racine, Phèdre
Unlike Shakespeare’s expansive historical sweep, Racine’s tragedies unfold in a single location over a short period, concentrating the emotional and moral conflict. His characters are often members of the aristocracy or historical figures, whose internal struggles are presented with a stark, almost clinical, precision. The tragic outcome is often a result of an internal moral failing or an uncontrollable passion, rather than external forces or a complex web of events. Racine’s work reflects a society that valued order, reason, and a clear understanding of human nature, even when depicting its most destructive aspects. Racine’s tragedies speak to a French society that, despite its pursuit of order, recognized the terrifying, almost inevitable, power of human passion to disrupt that order. They explore the moral and psychological consequences of defying strict social and religious codes, often within the confines of aristocratic life, where reputation and controlled emotion were paramount.
Divergent Stages, Shared Human Concerns: A Compelling Contrast
The comparison of these two dramatic traditions reveals fundamental differences in their artistic philosophies and their reflections of national character. English Renaissance drama, as seen in Marlowe and Shakespeare, was expansive, embracing complexity, psychological depth, and a vibrant, often chaotic, theatricality. It reveled in the individual’s boundless potential and tragic flaws, often breaking classical rules to achieve greater emotional impact and narrative freedom. The English stage was a mirror to a society undergoing rapid change, where human ambition and internal conflict were paramount, and where the individual’s journey, however tumultuous, was often the central focus.
French Neoclassical drama, in contrast, prioritized order, reason, and decorum. Molière’s comedies satirized social behaviors to uphold moral norms, while Racine’s tragedies meticulously dissected destructive passions within a tightly controlled framework. Their adherence to classical unities and their emphasis on elegant language reflected a desire for clarity, balance, and a more didactic approach to theatre. The French stage was a laboratory for examining universal human traits and societal structures, often through the lens of a single, dominant characteristic or emotion, emphasizing the importance of social harmony and rational control.
The most compelling statement arising from this comparison is that while English drama celebrated the unleashing of the individual, often leading to magnificent chaos, French drama sought to contain and analyze the individual within the strictures of reason and social order. The English stage, with its public accessibility and fewer formal constraints, became a crucible for exploring the raw, unvarnished human condition, reflecting a society more comfortable with its own contradictions and less centralized in its cultural authority. The French stage, often patronized by the monarchy and adhering to strict classical principles, became a refined instrument for social critique and the dissection of universal passions, reflecting a society that valued intellectual control, social hierarchy, and the triumph of reason over disruptive emotion.
Despite these significant stylistic and philosophical divergences, both traditions ultimately grappled with universal human concerns: ambition, love, betrayal, morality, and the search for meaning. Whether through the grand, sprawling narratives of Shakespeare and Marlowe, or the concentrated, analytical dramas of Molière and Racine, the theatre in both nations served as a vital arena for exploring the human condition, shaping national identities, and laying groundwork for future intellectual movements. The “stages of the soul” in the Renaissance and Neoclassical periods, though built on different principles, each offered profound insights into the timeless complexities of human nature.
THIS ESSAY WAS WRITTEN AND EDITED UTILIZING AI





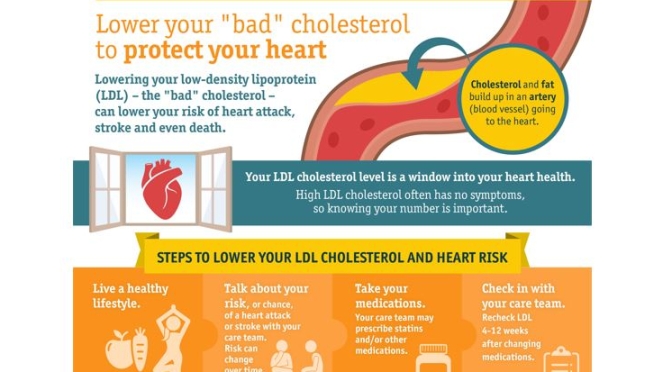


 These daily oscillations are controlled by the circadian clock, which is composed of an autoregulatory biochemical mechanism that is expressed in tissues throughout the body and is coordinated by a master pacemaker located in the suprachiasmatic nuclei of the brain (aka the SCN [
These daily oscillations are controlled by the circadian clock, which is composed of an autoregulatory biochemical mechanism that is expressed in tissues throughout the body and is coordinated by a master pacemaker located in the suprachiasmatic nuclei of the brain (aka the SCN [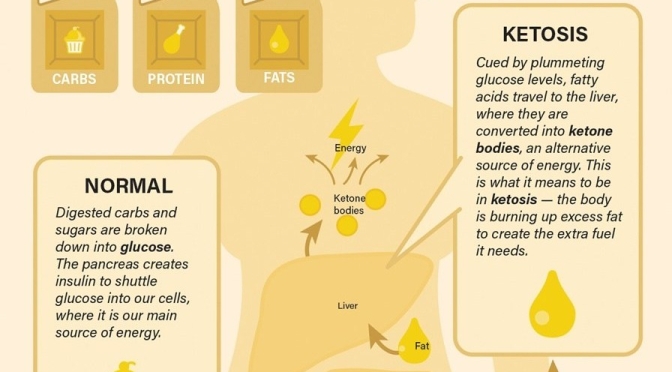
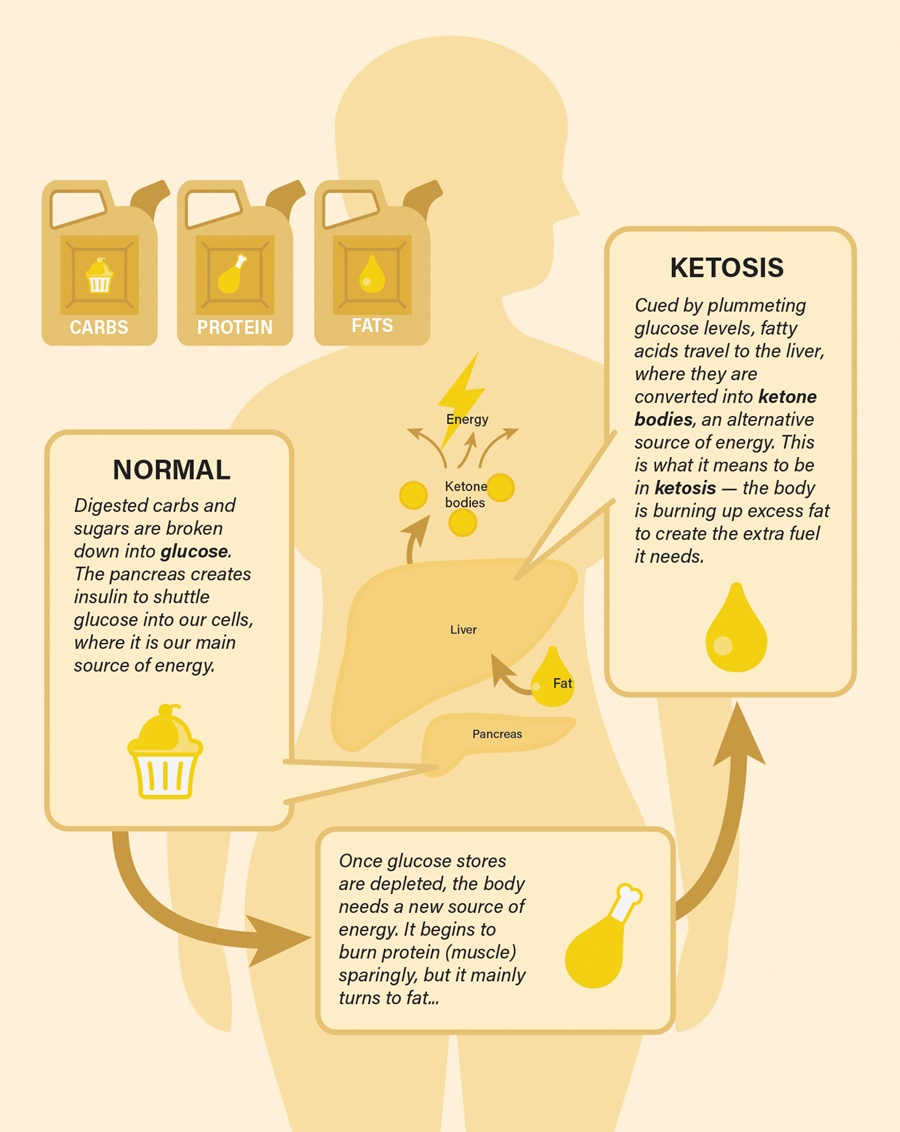
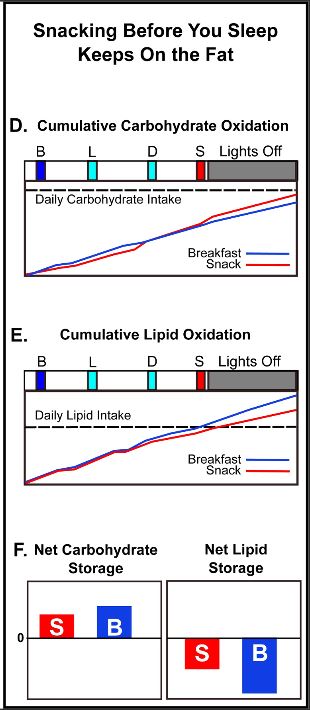
 to a paper Mattson and colleagues published in February in the experimental biology journal FASEB. In humans, fasting for 12 hours or more drops the levels of glycogen, a form of cellular glucose. Like changing to a backup gas tank, the body switches from glucose to fatty acids, a more efficient fuel. The switch generates the production of ketones, which are energy molecules that are made in the liver. “When the fats are mobilized and used to produce ketones, we think that is a key factor in accruing the health benefits,” says Mattson.
to a paper Mattson and colleagues published in February in the experimental biology journal FASEB. In humans, fasting for 12 hours or more drops the levels of glycogen, a form of cellular glucose. Like changing to a backup gas tank, the body switches from glucose to fatty acids, a more efficient fuel. The switch generates the production of ketones, which are energy molecules that are made in the liver. “When the fats are mobilized and used to produce ketones, we think that is a key factor in accruing the health benefits,” says Mattson.
 This latest paper builds on previous Newcastle studies supported by
This latest paper builds on previous Newcastle studies supported by 
 In summary, dietary fructose, but not glucose, supplementation of HFD impairs mitochondrial size, function, and protein acetylation, resulting in decreased fatty acid oxidation and development of metabolic dysregulation.
In summary, dietary fructose, but not glucose, supplementation of HFD impairs mitochondrial size, function, and protein acetylation, resulting in decreased fatty acid oxidation and development of metabolic dysregulation.