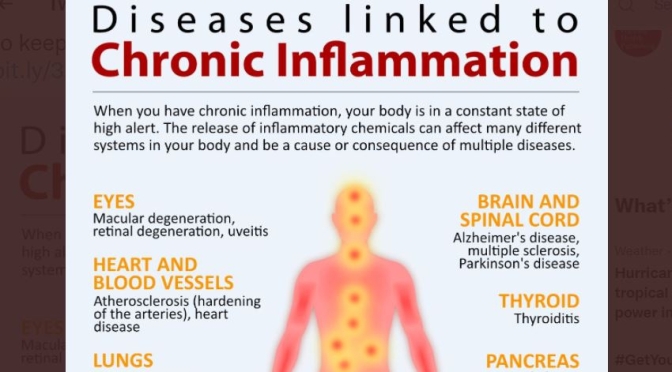
Scripps Research (April 21, 2023) – Chronic alcohol consumption may make people more sensitive to pain through two different molecular mechanisms—one driven by alcohol intake and one by alcohol withdrawal. That is one new conclusion by scientists at Scripps Research on the complex links between alcohol and pain.
The research, published in the British Journal of Pharmacology on April 12, 2023, also suggests potential new drug targets for treating alcohol-associated chronic pain and hypersensitivity.
“There is an urgent need to better understand the two-way street between chronic pain and alcohol dependence,” says senior author Marisa Roberto, PhD, the Schimmel Family Chair of Molecular Medicine, and a professor of neuroscience at Scripps Research. “Pain is both a widespread symptom in patients suffering from alcohol dependence, as well as a reason why people are driven to drink again.”
Alcohol use disorder (AUD), which encompasses the conditions commonly called alcohol abuse, alcohol dependence and alcohol addiction, affects 29.5 million people in the U.S. according to the 2021 National Survey on Drug Use and Health. Over time, AUD can trigger the development of numerous chronic diseases, including heart disease, stroke, liver disease and some cancers.