From iScience / Cell.com (June 26, 2020):
 …the beneficial effects of TRE are dose dependent, with greater reductions in body weight, fat mass, and improvement in glucose tolerance when a 9-h protocol was implemented versus 12 and 15 h. The optimal TRE time frame to recommend for people has not been tested. Clear improvements have been noted after 6-, 8-, 9-, and 10-h protocols. It is likely that the greater time restriction would result in greater weight losses, which may maximize the metabolic benefits.
…the beneficial effects of TRE are dose dependent, with greater reductions in body weight, fat mass, and improvement in glucose tolerance when a 9-h protocol was implemented versus 12 and 15 h. The optimal TRE time frame to recommend for people has not been tested. Clear improvements have been noted after 6-, 8-, 9-, and 10-h protocols. It is likely that the greater time restriction would result in greater weight losses, which may maximize the metabolic benefits.
Eating out of phase with daily circadian rhythms induces metabolic desynchrony in peripheral metabolic organs and may increase chronic disease risk. Time-restricted eating (TRE) is a dietary approach that consolidates all calorie intake to 6- to 10-h periods during the active phase of the day, without necessarily altering diet quality and quantity.
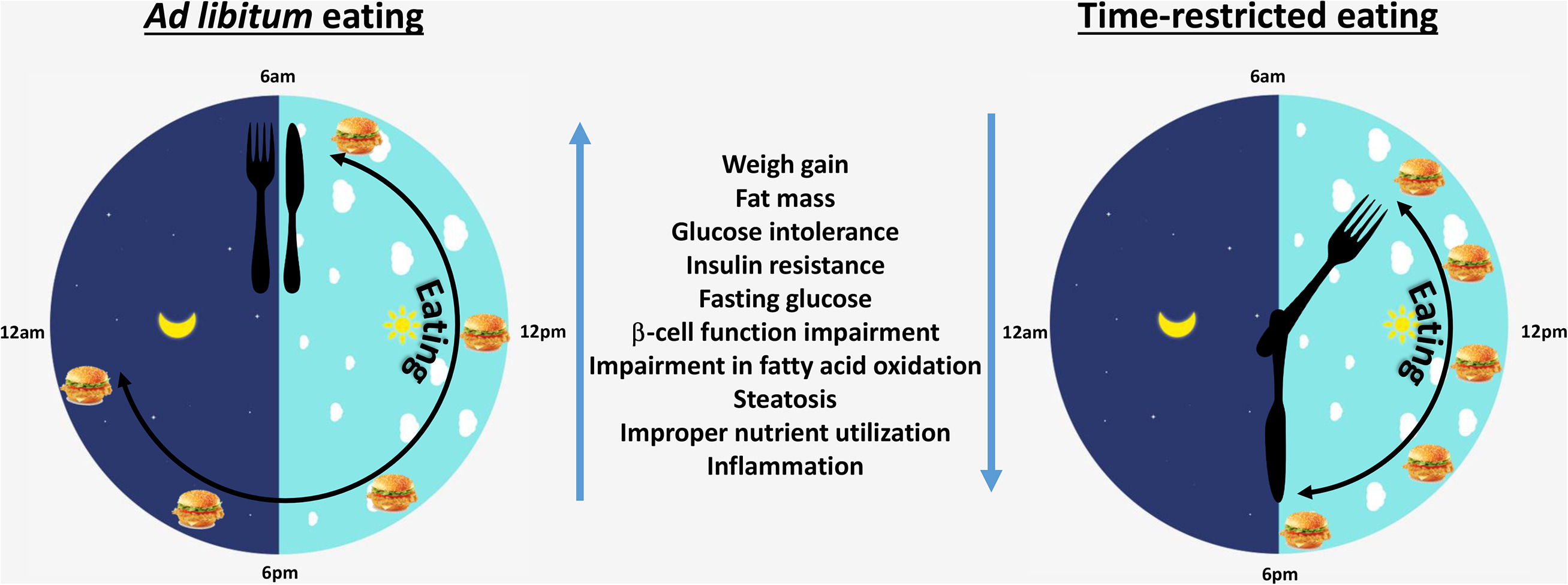
TRE reduces body weight, improves glucose tolerance, protects from hepatosteatosis, increases metabolic flexibility, reduces atherogenic lipids and blood pressure, and improves gut function and cardiometabolic health in preclinical studies. This review discusses the importance of meal timing on the circadian system, the metabolic health benefits of TRE in preclinical models and humans, the possible mechanisms of action, the challenges we face in implementing TRE in humans, and the possible consequences of delaying initiation of TRE.
Read full study









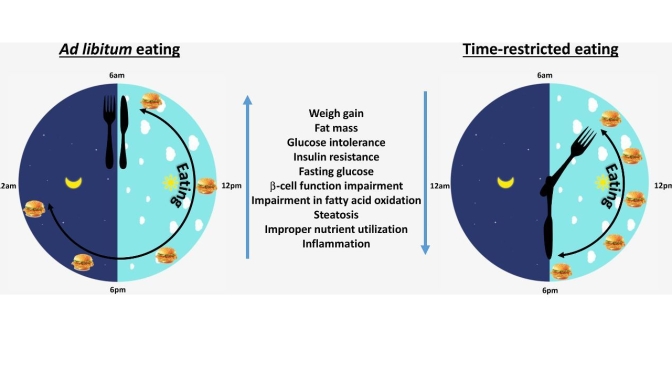
 …the beneficial effects of TRE are dose dependent, with greater reductions in body weight, fat mass, and improvement in glucose tolerance when a 9-h protocol was implemented versus 12 and 15 h. The optimal TRE time frame to recommend for people has not been tested. Clear improvements have been noted after 6-, 8-, 9-, and 10-h protocols. It is likely that the greater time restriction would result in greater weight losses, which may maximize the metabolic benefits.
…the beneficial effects of TRE are dose dependent, with greater reductions in body weight, fat mass, and improvement in glucose tolerance when a 9-h protocol was implemented versus 12 and 15 h. The optimal TRE time frame to recommend for people has not been tested. Clear improvements have been noted after 6-, 8-, 9-, and 10-h protocols. It is likely that the greater time restriction would result in greater weight losses, which may maximize the metabolic benefits.

 “What you want is more, small fat cells rather than fewer, large fat cells,” Jackson said. “A large fat cell is not a healthy fat cell. The center is farther away from an oxygen supply, it sends out bad signals and it can burst and release toxic contents.” Large fat cells are associated with insulin resistance, diabetes and inflammation, he added.
“What you want is more, small fat cells rather than fewer, large fat cells,” Jackson said. “A large fat cell is not a healthy fat cell. The center is farther away from an oxygen supply, it sends out bad signals and it can burst and release toxic contents.” Large fat cells are associated with insulin resistance, diabetes and inflammation, he added.