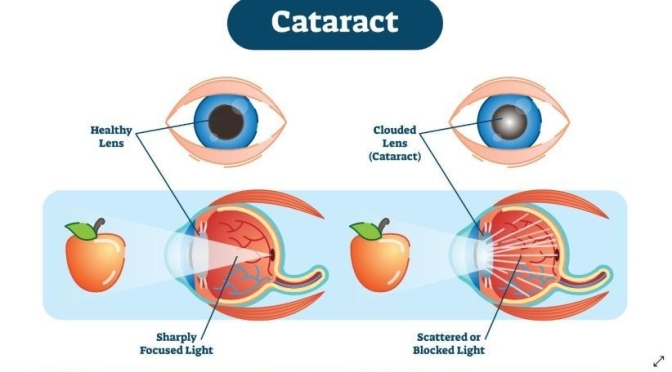
Cataract surgery is one of the safest and most effective types of surgery. In about 90 percent of cases, people who have cataract surgery have better vision afterward. Learn more about aging and your eyes at: https://www.nei.nih.gov/learn-about-e….
Tag Archives: NIH
Covid-19 Infographic: ‘The Journey Of A Vaccine’ (NIH)


Health Update: The First Clinical Trial Of A “Coronavirus / Covid-19” Vaccine Begins (NIH)
From the National Institutes of Health (NIH) (March 17, 2020):
 “Finding a safe and effective vaccine to prevent infection with SARS-CoV-2 is an urgent public health priority,” said NIAID Director Anthony S. Fauci, M.D. “This Phase 1 study, launched in record speed, is an important first step toward achieving that goal.”
“Finding a safe and effective vaccine to prevent infection with SARS-CoV-2 is an urgent public health priority,” said NIAID Director Anthony S. Fauci, M.D. “This Phase 1 study, launched in record speed, is an important first step toward achieving that goal.”
The vaccine is called mRNA-1273 and was developed by NIAID scientists and their collaborators at the biotechnology company Moderna, Inc., based in Cambridge, Massachusetts. The Coalition for Epidemic Preparedness Innovations (CEPI) supported the manufacturing of the vaccine candidate for the Phase 1 clinical trial.
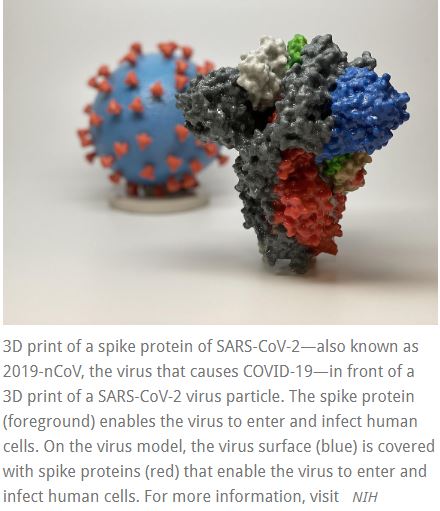 A Phase 1 clinical trial evaluating an investigational vaccine designed to protect against coronavirus disease 2019 (COVID-19) has begun at Kaiser Permanente Washington Health Research Institute (KPWHRI) in Seattle. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, is funding the trial. KPWHRI is part of NIAID’s Infectious Diseases Clinical Research Consortium. The open-label trial will enroll 45 healthy adult volunteers ages 18 to 55 years over approximately 6 weeks. The first participant received the investigational vaccine today.
A Phase 1 clinical trial evaluating an investigational vaccine designed to protect against coronavirus disease 2019 (COVID-19) has begun at Kaiser Permanente Washington Health Research Institute (KPWHRI) in Seattle. The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, is funding the trial. KPWHRI is part of NIAID’s Infectious Diseases Clinical Research Consortium. The open-label trial will enroll 45 healthy adult volunteers ages 18 to 55 years over approximately 6 weeks. The first participant received the investigational vaccine today.
The study is evaluating different doses of the experimental vaccine for safety and its ability to induce an immune response in participants. This is the first of multiple steps in the clinical trial process for evaluating the potential benefit of the vaccine.
Top New Science Podcasts: NIH Grant Diversity, Post-Traumatic Stress Memory Suppression (ScienceMag)
 On this week’s show, senior correspondent Jeffrey Mervis joins host Sarah Crespi to discuss a new National Institutes of Health (NIH) grant program that aims to encourage diversity at the level of university faculty with the long-range goal of increasing the diversity of NIH grant recipients.
On this week’s show, senior correspondent Jeffrey Mervis joins host Sarah Crespi to discuss a new National Institutes of Health (NIH) grant program that aims to encourage diversity at the level of university faculty with the long-range goal of increasing the diversity of NIH grant recipients.
Sarah also talks with Pierre Gagnepain, a cognitive neuroscientist at INSERM, the French biomedical research agency, about the role of memory suppression in post-traumatic stress disorder. Could people that are better at suppressing memories be more resilient to the aftermath of trauma?
Science Podcasts: NIH Scientist Loan Repayment Conflicts, Undersea Cables As Seismic Sensors
The National Institu tes of Health’s largest loan repayment program was conceived to help scientists pay off school debts without relying on industry funding. But a close examination of the program by investigative correspondent Charles Piller has revealed that many participants are taking money from the government to repay their loans, while at the same time taking payments from pharmaceutical companies. Piller joins Host Sarah Crespi to talk about the steps he took to uncover this double dipping and why ethicists say this a conflict of interest.
tes of Health’s largest loan repayment program was conceived to help scientists pay off school debts without relying on industry funding. But a close examination of the program by investigative correspondent Charles Piller has revealed that many participants are taking money from the government to repay their loans, while at the same time taking payments from pharmaceutical companies. Piller joins Host Sarah Crespi to talk about the steps he took to uncover this double dipping and why ethicists say this a conflict of interest.
Sarah also talks with Nate Lindsey, a Ph.D. candidate at the University of California, Berkeley, about turning a 50-meter undersea fiber optic cable designed to move data into a sensor for activity in the ocean and the land underneath. During a 4-day test in Monterey Bay, California, the cable detected earthquakes, faults, waves, and even ocean-going storms.
For this month’s books segment, Kiki Sandford talks with Dan Hooper about his book At the Edge of Time: Exploring the Mysteries of Our Universe’s First Seconds.